Data Management
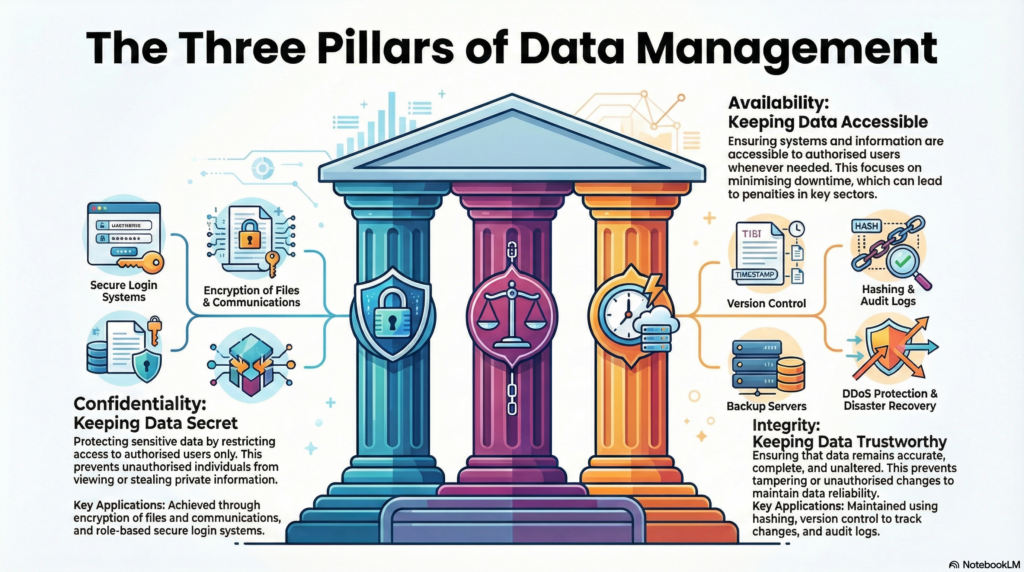
Data integrity is the backbone of reliable science, ensuring that information remains accurate, complete, and trustworthy. It prevents tampering or unauthorised changes using rigorous verification techniques to maintain reliability.
Real-life applications include utilising checksums and hashes to verify data during transmission, employing version control systems to track changes, and analyzing audit logs to detect unauthorised activity.
The TRANSCEND™ Digital Suite is built to master these challenges in the pharmaceutical sector. It eliminates manual data entry errors by automatically capturing and synchronising data to ensure absolute consistency and integrity . The suite reinforces security through an Authority Access Matrix for unique user logins, electronic signatures, and robust audit trails. Furthermore, it adheres to strict compliance standards like eg CFR 21 Part 11, ensuring your data remains valid and compliant.
Ready to secure your lab’s data? Reach out to us to schedule a demonstration.